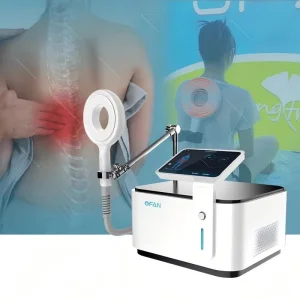
California Series: Advanced Neck Pain Relief PEMF Magnetic Therapy Medical Equipment
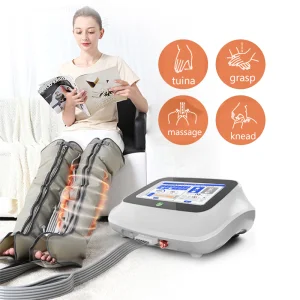
Texas Rehabilitation Grade: Air Pressure Therapy Device for Full Body Pain Relief

New York Professional Class IV High Power Laser Therapy 980nm 1064nm Multifunction Device

Florida Wellness Edition: TDP Far Infrared Heat Lamp Therapy for Home & Clinic Use
The United States stands at the global forefront of medical device innovation, particularly in the sector of Pain Relief Therapy Devices. With an aging population and a significant shift away from opioid-based treatments, the demand for non-invasive, drug-free pain management solutions has reached an all-time high. Manufacturers across hubs like Minnesota’s "Medical Alley," California’s Silicon Valley, and Massachusetts are redefining how chronic pain is treated through bio-electronic medicine and advanced physiotherapy technologies.
In the United States, pain relief devices are strictly regulated by the FDA. Most therapeutic devices, such as TENS units, PEMF machines, and Class IV Lasers, fall under Class II medical devices. This ensures that every factory operating within or supplying to the US market adheres to stringent Quality System Regulations (QSR) and ISO 13485 standards, guaranteeing patient safety and clinical efficacy.
The industrial status of this sector is characterized by a heavy investment in R&D. American manufacturers are increasingly integrating Artificial Intelligence (AI) and IoT connectivity into therapy devices. This allows for personalized treatment protocols where the device adjusts its intensity based on real-time biofeedback from the patient. This "Smart Therapy" trend is transforming traditional clinics into data-driven recovery centers.
The shift toward Non-Opioid Pain Management is the single most significant driver for the industry in the United States. Following the national health crisis related to prescription painkillers, both the government and private healthcare providers are incentivizing the use of medical devices. Here are the key technologies currently dominating the US factories and clinics:
Pulsed Electromagnetic Field therapy is gaining massive traction in US sports medicine and orthopedic clinics for bone healing and deep tissue repair.
Class IV lasers are now the standard for rapid inflammation reduction and accelerated cellular regeneration in professional athletic training rooms.
Combining thermal and mechanical energy, these devices are essential for treating chronic tendinopathies and myofascial pain syndromes.
Furthermore, Localized Application Scenarios in the US are diversifying. While hospitals remain a primary market, there is a surge in "Boutique Recovery Labs" and "Longevity Centers" in cities like Los Angeles, Miami, and Chicago. These facilities offer high-end therapy sessions using medical-grade equipment for bio-hacking and performance optimization. Simultaneously, the "Home-Health" movement is driving manufacturers to miniaturize professional technology into portable, user-friendly devices for at-home chronic pain management.
Hangzhou StavroCare Medical Co., Ltd. is a professional manufacturer specializing in advanced physiotherapy equipment and health assessment systems. With a strong focus on non-invasive therapy solutions, the company integrates research, development, production, and global distribution to deliver reliable medical technologies for modern healthcare needs.
StavroCare is committed to innovation in rehabilitation and preventive healthcare. Its product portfolio includes physiotherapy devices, microcirculation improvement systems, magnetic therapy equipment, and intelligent health assessment platforms. Designed for hospitals, rehabilitation centers, and home care environments, these solutions aim to enhance treatment efficiency, improve patient outcomes, and support long-term wellness management.

Backed by an experienced engineering team and strict quality control standards, StavroCare ensures that every product meets international performance and safety requirements. The company continuously invests in advanced manufacturing technologies and digital healthcare integration, enabling smarter and more connected medical solutions.
With a customer-oriented approach, Hangzhou StavroCare Medical Co., Ltd. provides OEM and ODM services tailored to global partners, helping brands bring competitive and innovative healthcare products to market. Dedicated to quality, reliability, and continuous improvement, StavroCare is building its reputation as a trusted supplier in the global non-invasive medical device industry.

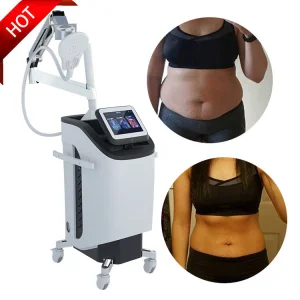
Chicago Professional: Laser Magnetic Therapy Device for Back Pain & Physical Therapy

Seattle Home-Care: Electric Potential Therapy Device for Migraine & Headache Relief

Boston Clinical Grade: My-S008j Spectrum Therapy Device for Pain Relief Centers
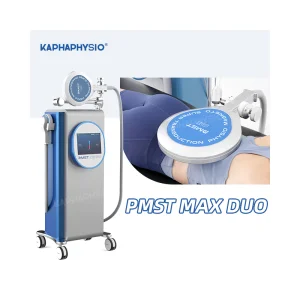
Houston High-Tech: Advanced 7 Tesla EMF Therapy Device for Effective Pain Relief

Denver Portable: Pakiss 660nm 850nm Red Light Therapy Device for Whole Body Care

Atlanta Sports Med: Professional CET RET Tecar Therapy Physiotherapy Device
Phoenix Recovery: High Power LLLT Cold Laser Therapy Pain Relief Device
Las Vegas Clinic: Portable PEMF Therapy Device for Rapid Muscle Recovery
As we look toward the next decade, the United States pain relief therapy device market is expected to integrate even more deeply with digital health ecosystems. We are seeing the rise of "Therapy-as-a-Service," where manufacturers provide not just the hardware, but a cloud-based platform that tracks patient progress and optimizes treatment parameters remotely. This is particularly vital for the rural US population, where access to specialized physical therapy clinics may be limited.
Factories are also focusing on Sustainability and Ergonomics. Modern American clinicians demand devices that are not only effective but also energy-efficient and easy to operate during long shifts. The integration of haptic feedback and voice-controlled interfaces is currently in the prototype stage at many leading US R&D labs.
Choosing the right manufacturer involves evaluating their clinical trial history and their commitment to post-market surveillance. For US-based distributors, partnering with a factory like StavroCare ensures a supply chain that understands the nuances of the American medical landscape, from FDA documentation to the specific voltage and safety requirements of US medical facilities.
In conclusion, the United States remains the most dynamic market for Pain Relief Therapy Devices. By combining local industrial expertise with global manufacturing efficiency, providers can offer patients the highest standard of care, moving the needle toward a future where chronic pain is managed safely, effectively, and without the risks associated with traditional pharmacology.