Pharmaceutical Oncology Drugs Formulation System
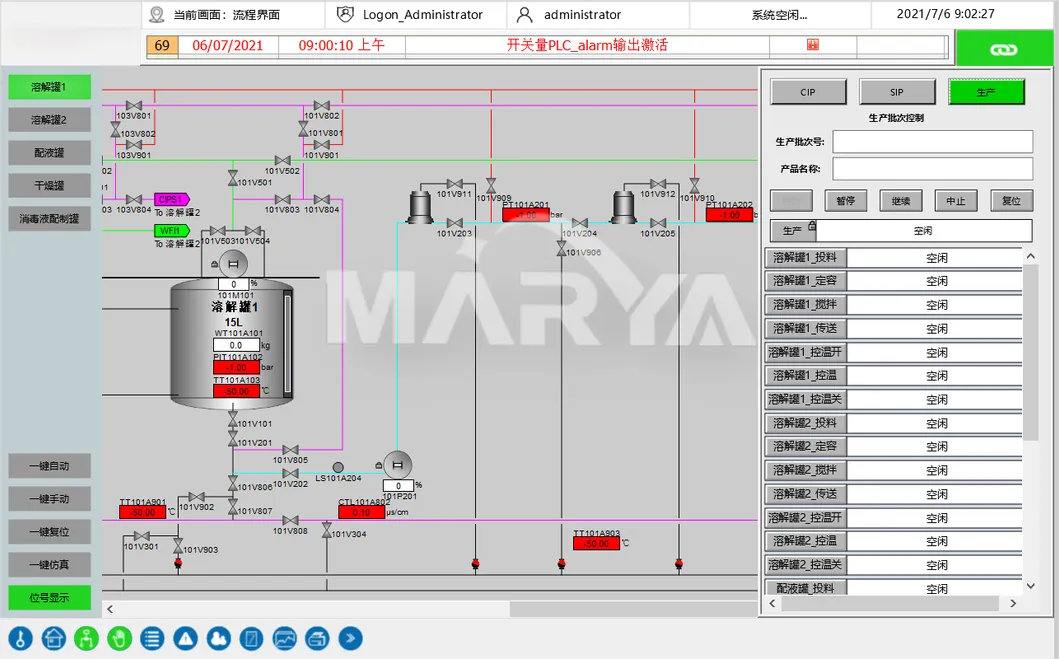
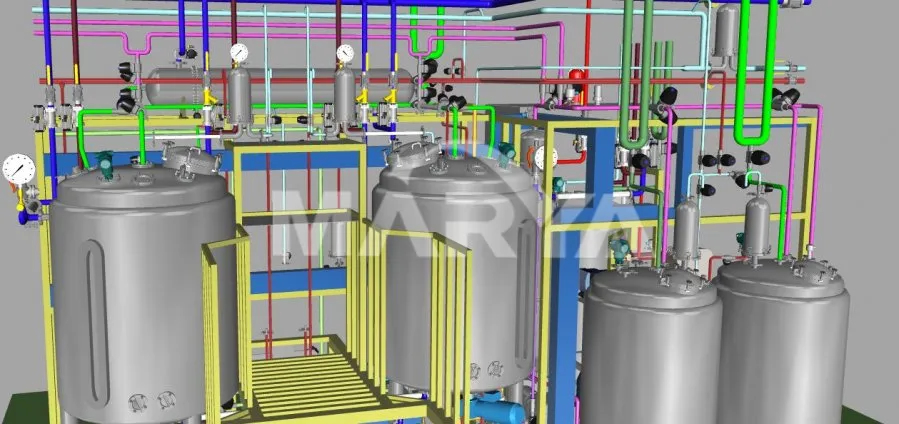
The Formulation System is a specialized pharmaceutical apparatus designed for liquid mixing and dispensing operations. This equipment enables the preparation of pharmaceutical solutions by combining active pharmaceutical ingredients (APIs) with solvents such as water for injection. Through integrated processes including controlled stirring, heating, sterilization, and filtration, the system produces medicinal solutions at precise target concentrations.
Basic Information
Certification
CE, FDA, GMP, ISO
Control System
Fully Automatic
Treatment Process
Filtration, Ultrafiltration
Tank Material
Stainless Steel 316L
Core Advantages
Modular Design & Automation
Pre-tested modular equipment reduces installation costs and ensures flexibility. The system features 1-click CIP/SIP with auto-detection and computerized recipe management with audit trails.
Real-Time Monitoring & 3D Modeling
Critical parameter control for temperature, DO, and pH with auto-alerts. Pre-production 3D modeling optimizes piping and valve placement for ergonomic operation.
Frequently Asked Questions
1. What materials are used in the construction of the formulation tanks?
The system utilizes high-grade 316L stainless steel for all product-contact surfaces, ensuring maximum corrosion resistance and compliance with FDA and GMP standards.
2. Does the system support automated cleaning and sterilization?
Yes, the system is equipped with a fully automated 1-click CIP (Clean-in-Place) and SIP (Sterilize-in-Place) function, including automatic detection for cycle completion.
3. What is the range of working volumes available?
The system is highly scalable, with working volumes ranging from 2 Liters for R&D purposes up to 15,000 Liters for large-scale industrial production.
4. How precise is the temperature and stirring control?
The system provides high-precision control with a temperature accuracy of ±0.2ºC and stirring speed accuracy of ±3.0% within a range of 0~450rpm.
5. Is the control system compliant with digital record requirements?
Yes, the computerized system includes recipe management, electronic signatures, electronic records, and a full audit trail to meet modern pharmaceutical regulatory requirements.
6. Can the system handle biological products like vaccines?
Absolutely. We offer a specialized Biological Formulation System specifically designed for antibodies, vaccines, blood products, and recombinant proteins.